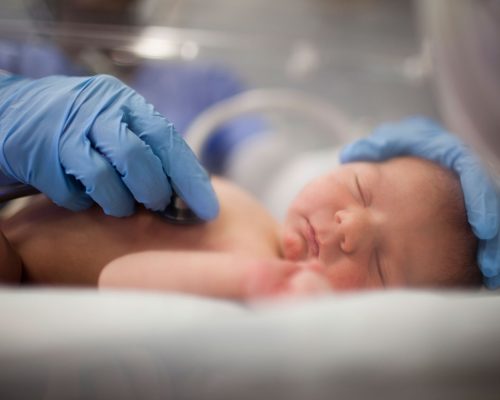
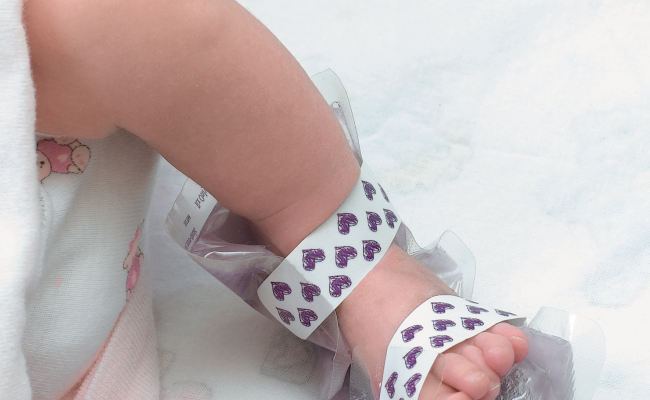
From a high risk delivery to a NICU graduation
Our infant care solutions are with you every step of the way.
Areas of Care
From a high risk delivery to a NICU graduation and everything in between, International Biomedical’s infant care solutions are with you every step of the way.
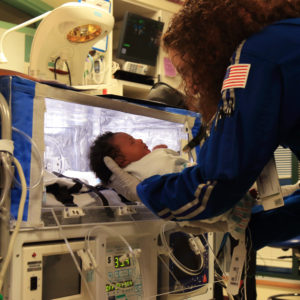
Neonatal Transport
Specialized neonatal medical equipment is mission critical to a successful transport. We offer a wide range of medical devices and neonatal transport accessories designed to support the infant’s clinical needs during ground or aeromedical transport.
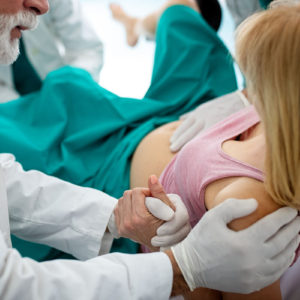
Labor & Delivery
Entering a new world can be tough, and a seamless transition into life is the goal for every birth. Our L&D solutions provide unique and novel clinical features which focus on care for the mother & her baby.
NICU & Nursery
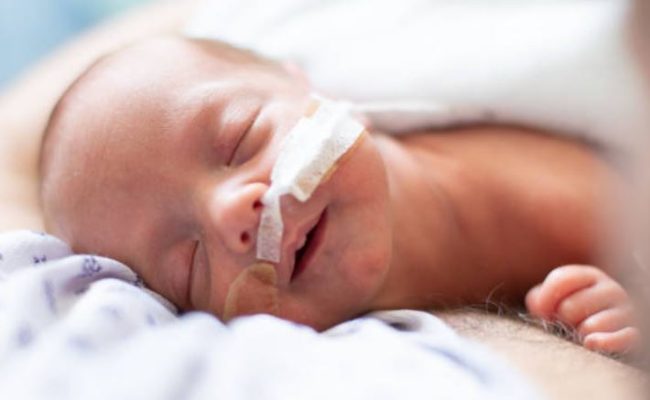
The needs of a NICU or Special Care Nursery patient are unique and wide ranging in complexity. From a simple swaddle to an advanced radiant warmer, our NICU & Nursery solutions nurture the infant and provide life saving therapies and treatments with a delicate touch.
Neuro NICU
There is nothing more fragile or critical than a developing brain. From diagnosis to treatment of a brain injury, our Neuro NICU solutions provide critical insight and protective support for a very precious package.
Developmental Care
Developmental care is more than just a concept for International Biomedical, it’s a design principle. Our developmental care solutions nurture the infant and provide life saving treatment with a delicate touch.
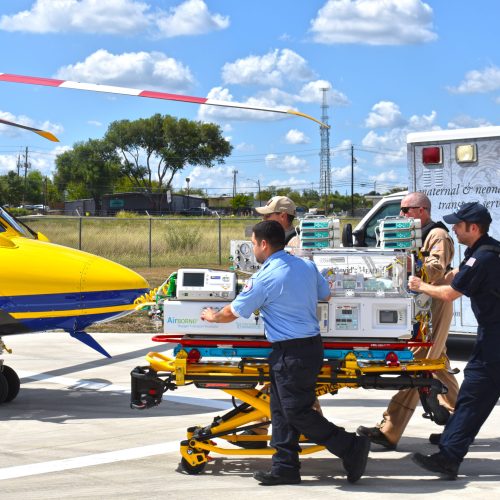
Neonatal Transport
Transporting a critically ill infant requires unique equipment and highly trained clinicians to accomplish the live saving mission. Specialized neonatal medical equipment is mission critical to a successful transport. We offer a wide range of medical devices and neonatal transport accessories designed to support the infant’s clinical needs during ground or aeromedical transport.
Labor & Delivery
Entering a new world can be tough, and a seamless transition into life is the goal for every birth. Our L&D solutions provide unique and novel clinical features which focus on care for the mother & her baby.
NICU & Nursery
The needs of a NICU or Special Care Nursery patient are unique and wide ranging in complexity. From a simple swaddle to an advanced radiant warmer, our NICU & Nursery solutions nurture the infant and provide life saving therapies and treatments with a delicate touch.
Neuro NICU
There is nothing more fragile or critical than a developing brain. From diagnosis to treatment of a brain injury, our Neuro NICU solutions provide critical insight and protective support for a very precious package.
Developmental Care
Developmental care is more than just a concept for International Biomedical, it’s a design principle. Our developmental care solutions nurture the infant and provide life saving treatment with a delicate touch.
Equip your team with the Best Products
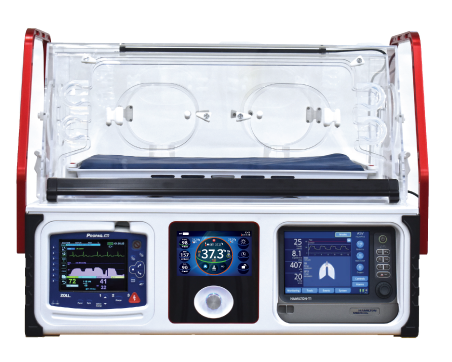

Transport Incubators
AirBorne Transport Incubators have been a fixture in the infant transport world for the last 45 years. Whether it is a critical aeromedical transport or a simple back transport of a healthy infant, AirBorne Transport incubators are customizable for any mission.

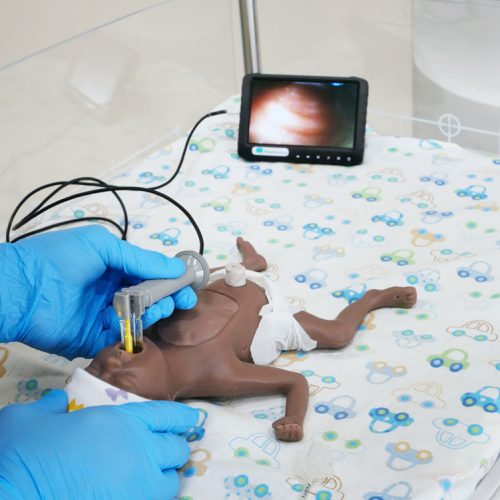
Video Intubation System
The NeoView Neonatal Video Intubation System enables videolaryngoscopy for tracheal intubation on the most fragile patients.

Infant Positioning Aid
The Transportle Neuro Protection system is a clinically advanced positioning aid that provides protective support for the most precious package during transport and into the NICU.
Early Registration OPEN!
20th Annual AirBorne Neonatal & Pediatric Transport Conference
Austin, Texas | May 8-10, 2024

Infant Cooling System
The small and lightweight Tecotherm Neo offers total body infant cooling and warming for your mobile therapeutic hypothermia needs.

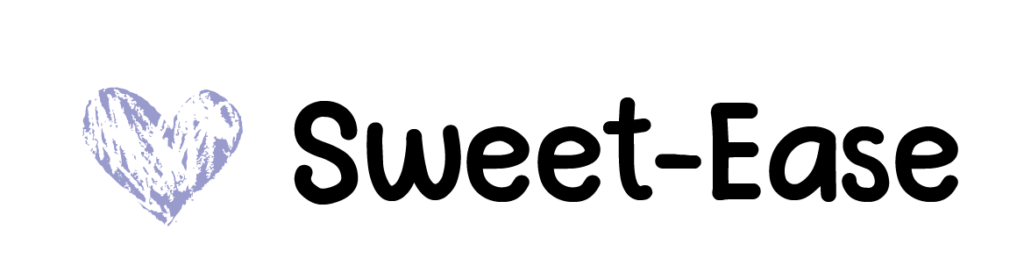
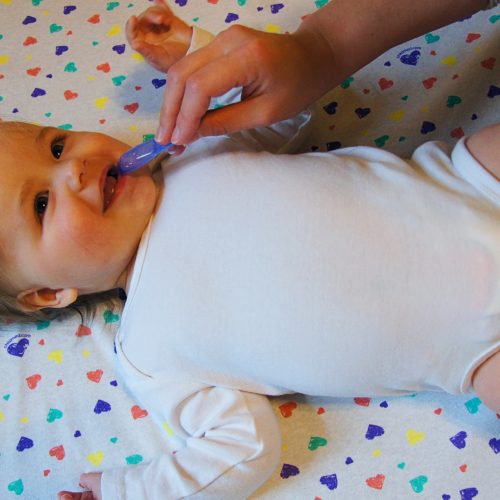
Sucrose Solution
Sweet-Ease 24% sucrose and purified water solution is used to soothe the baby and is available in 15 mL cups or a 2 mL vial.

Fiber Optic Phototherapy
The BiliCocoon fiber optic phototherapy system provides a soft and flexible phototherapy treatment option that is compatible with warmers, incubators and kangaroo care.
The Technology You Need
When It Matters Most
Browse Top Care Areas
From a high risk delivery to a NICU graduation and everything in between, International Biomedical’s infant care solutions are with you every step of the way.

Find Your Regional Sales & Clinical Reps
Innovating
Neonatal and Perinatal Products and Technologies Worldwide For Over 45 years
International Biomedical Ltd. is an Austin, Texas based company that designs, manufactures, and distributes innovative neonatal and perinatal products and technologies to hospitals worldwide. For over 45 years, our mission has been to provide the best possible outcomes for the newborn infant, the critically ill, and their families. Our family owned and operated company stands 100+ strong, spans the globe, and promotes a culture of respect and integrity. While manufacturing quality products is our top priority, we are also committed to giving back to our local communities and reducing our impact on the environment.



Connect With Us
For more information about our family of products, please contact us:
-
8206 Cross Park Drive
Austin, Texas 78754 -
1.800.433.5615
-
sales@int-bio.com